omerguul: #chemistry

#chemistry
kemistig: Oh! Shiny molecule! Potassium tert-butoxide (t-BuOK)...
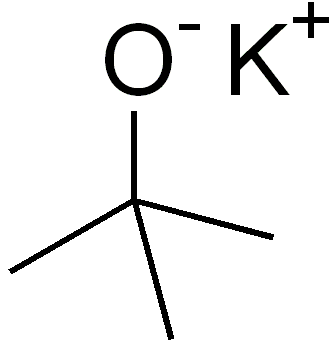
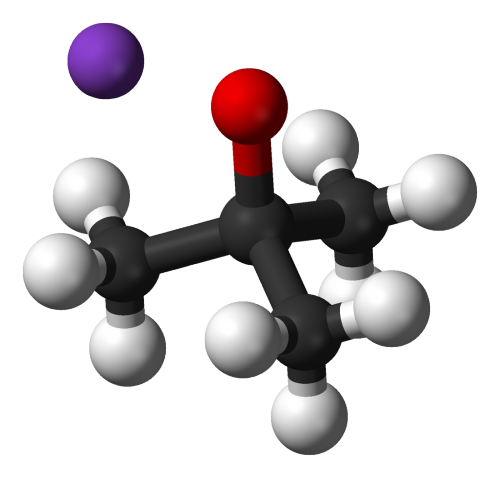
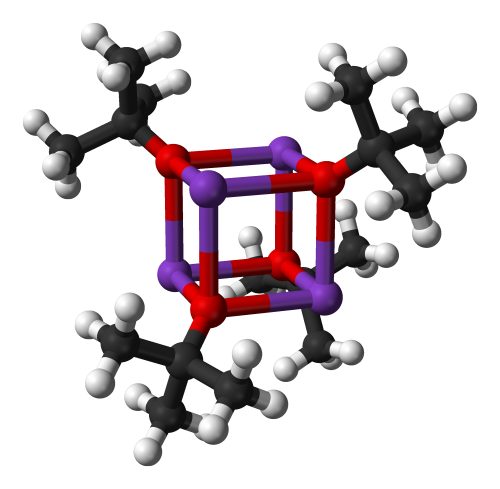
Oh! Shiny molecule!
Potassium tert-butoxide (t-BuOK) is a convenient strong base created when potassium metal reacts with tertiary butanol. A fun thing about this compound is the cubic tetramer it forms upon sublimation - it apparently exists even in gas phase!
Due to its (quite visible) steric hindrance it is non-nucleophilic, which makes it a good choice for many syntheses, but its basicity is somewhat limited with a conjugate acid pKa of 17.
Another interesting (or annoying) property is that it tends to form the Hoffmann elimination product, contrary to the "rule" that proposes the most substituted alkene is formed. (Further reading on this blog!)
elvenwanderer06: Again, chemistry can be so beautiful.



Again, chemistry can be so beautiful.
selenocysteine: Recrystallization via the layered solvent...

Recrystallization via the layered solvent method! The bottom layer is DCM with an iridium complex dissolved inside, and the top layer is ether. The complex is soluble in DCM but not very soluble in ether, so hopefully we'll get some crystals within a day or two as the layers diffuse into each other. :D
double-o-socks: Just makin quantum dots because chemistry is my...




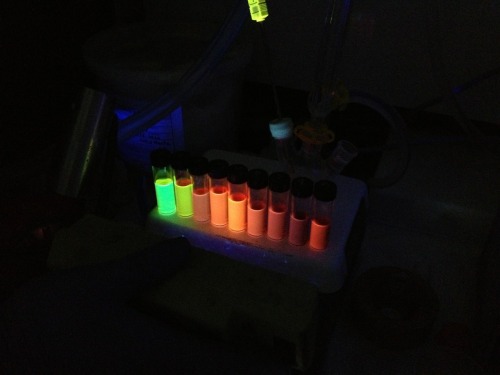
Just makin quantum dots because chemistry is my life right now
droppingthephysics: Refraction causes a rigid object to appear...


Refraction causes a rigid object to appear fragmented when it is placed in two different optical mediums.
Photo

adventures-in-the-lab: The Big Dipper Image Credit &...
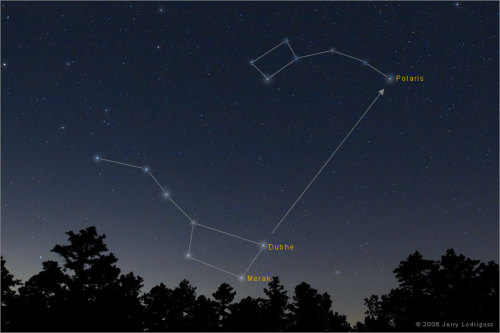
The Big Dipper
Image Credit & Copyright: Jerry Lodriguss (Catching the Light)Explanation: Do you see it? This common question frequently precedes the rediscovery of one of the most commonly recognized configurations of stars on the northern sky: the Big Dipper. This grouping of stars is one of the few things that has likely been seen, and will be seen, by every generation. The Big Dipper is not by itself a constellation. Although part of the constellation of the Great Bear (Ursa Major), the Big Dipper is an asterism that has been known by different names to different societies. Five of the Big Dipper stars are actually near each other in space and were likely formed at nearly the same time. Connecting two stars in the far part of the Big Dipper will lead one to Polaris, the North Star, which is part of the Little Dipper. Relative stellar motions will cause the Big Dipper to slowly change its apparent configuration over the next 100,000 years.
A very visible constellation, even in the suburbs where I live. I try to find the North Star every time the skies are clear =)
atomicallena: Argon Ice In the video above, argon is liquified...
Argon Ice
In the video above, argon is liquified and frozen (wut), using liquid nitrogen, ultimately producing argon ice (double wut). I think it's quite interesting to think of a solid noble gas.
At room temperature, argon exists as a gas. Argon has a melting point of -186.36 °C (-303.448 °F) and a boiling point of -185.847 °C (-302.525 °F) . The narrow range between the melting and boiling points is demonstrated around 4:37 when the drops of liquid argon immediately boil as it lands on the surface.
Fun fact: argon is the third most abundant gas (0.93%) in the earth's atmosphere, behind nitrogen (78.09%) and oxygen (20.95%).
Sources used: "Argon Ice" Video and PubChem
fuckyeahlabglass: vacuum & volumetric flasks on the drying...


vacuum & volumetric flasks on the drying rack
foadgreen: Some of our inorganic crystal creations in the lab.



Some of our inorganic crystal creations in the lab.
Spectral Database for Organic Compounds
Spectral Database for Organic Compounds:For anyone taking organic chem, this site saved my life. Particularly helpful when identifying unknown compounds based on H NMR and IR Spectra
Used this all the time. Great resource =)
clothesb4bros: "Lewisite, in chemical warfare, poison blister...

"Lewisite, in chemical warfare, poison blister gas developed by the United States for use during World War I. Chemically, the substance is dichloro(2-chlorovinyl)arsine, a liquid whose vapour is highly toxic when inhaled or when in direct contact with the skin. It blisters the skin and irritates the lungs. Any part of the body that is contacted by the liquid or vapour suffers inflammation, burns, and tissue destruction."—Encyclopedia Britannica
fuckyeahlabglass: Distillation and reflux heads

Distillation and reflux heads
-nerdglasses: For chem people: This is only 25 μM. Crazy. For...

For chem people: This is only 25 μM. Crazy. For all people: the photo really doesn't do the color justice. Definitely the prettiest solution I've ever made in #APchem #chemistry #pretty #science #nofilter @akn1128
Found this in some old papers of mine. It's a fun little...
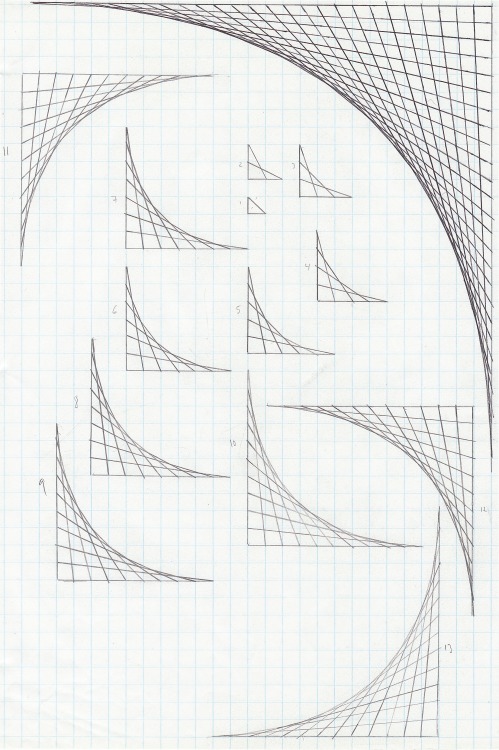
Found this in some old papers of mine. It's a fun little exercise you can try using some graph paper and a ruler.
Easy, looks cool, and gives a bit of insight into tangents.
biological-chemistry: The rate of reaction increases with the...
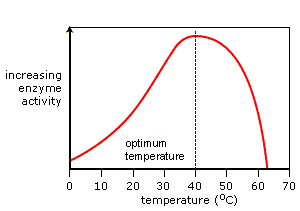
The rate of reaction increases with the temperature. However extreme temperature denatures the tertiary structure of the enzyme. Starting at zero human enzymes show increasing temperature, reaching an optimum activity at around 37 temperatures after which they denature. Destroying the proteins active sites.
Sodium Lamp: I REALLY NEED HELP
Sodium Lamp: I REALLY NEED HELP:can someone please please explain to me in laymens terms what Linus Pauling did to win his nobel prize in Chemistry!
Pauling won the 1954 Nobel Prize in Chemistry for his work on elucidating the chemical bond. His work is summarized in his 1939 book The Nature of…
danlotr: Having fun with coloured flames in Chemistry :)

Having fun with coloured flames in Chemistry :)
adventures-in-the-lab: Sieving silica sieves from biomass ash Burning biomass for heat and power...
Sieving silica sieves from biomass ash
Burning biomass for heat and power could produce as much as 2000 TWh by 2020, which would produce 4–15.6 million tonnes of waste ash, per year, in Europe alone. To address the problem of what to do with all this waste, scientists in the UK have developed a method to convert this ash into mesoporous silica.














Комментариев нет:
Отправить комментарий